Professor Sharon Peacock is a world-renowned expert in tracking bacterial antibiotic resistance genes using molecular techniques (1). She reported to the UK Parliament last week that a finger prick test kit for anti-COVID-19 antibodies will soon be available. Sharon, who was a Trustee of Antibiotic Research UK until she resigned last year to take up the role of Director of Infection Control at Public Health England, told MPs on the Science and Technology Committee that evaluation of the kits would be completed within days (2) and that mass screening could be possible in days.
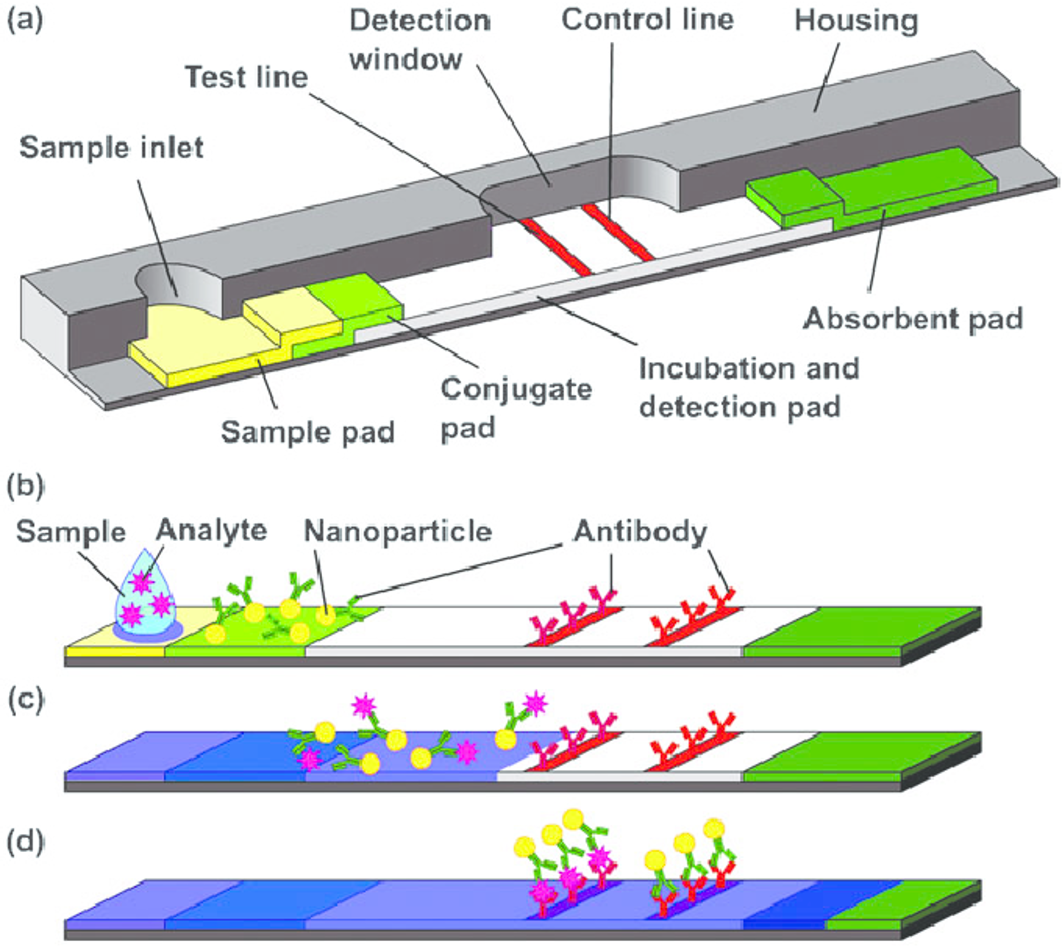
The Government appeared to take a more cautious approach to the announcement, saying that the tests would not be available so quickly. As I wrote in my blog of 23 March 2020, if we don’t test people quickly then we won’t know who has had a COVID-19 infection. There are two types of test we need. The type that Professor Peacock described, which tells us if a person has had an infection (see https://www.assaygenie.com/rapid-covid19-antibody-detection-tests-principles-and-methods) for more details , and another type that tells us if a person is carrying the COVID-19 virus. The latter is important because there may be as many as 30% of virus carriers who show no symptoms (see future blog on silent carriers).
The first group of people who should be tested are NHS staff treating COVID-19 patients. In some hospitals up to 50% of the staff are off sick and having to self-isolate (3). This means that our brilliant frontline NHS staff are exposed unnecessarily doing their jobs. A lack of Personal Protective Equipment (PPE) for NHS staff seems a major contributory factor.
The danger with a rapidly introduced kit is that it will not have been thoroughly evaluated. Antibody-based kits of the type described are similar to pregnancy testing kits. Millions, if not billions, of the latter have been manufactured over the past 25 years or so – their failure rate is around 1%. When any new test is introduced, the question always asked is what are the percentage failure rates? I.e. a false positive result occurs when the person may not have been infected with COVID-19. Or false negative rates where the person has been infected, is immune, but the test fails to pick this up. This means even though there is a lot of pressure to roll out kits quickly, they must be reliable. Testing for reliability takes time; time we do not have.
Please remember to follow government guidance on coronavirus at all times. Remember, evidence shows that secondary bacterial infections and subsequent sepsis are a common cause of death in people with coronavirus.
References
(1) Simon R Harris, Edward J PCartwright, EstéeTörök, Matthew T G Holden, Nicholas M Brown, Amanda LOgilvy-Stuart, Matthew J Ellington, Michael A Quail, Stephen D Bentley, JulianParkhill, Sharon J Peacock (2013) Whole-genome sequencing for analysis of an outbreak of methicillin-resistant Staphylococcus aureus: a descriptive study, Lancet Inf Dis, 13: 130–36